
Fernando Augusto de Souza, Ph.D.
Technician Specializing in Monogastric, ICC Brazil
Protein is the heaviest nutrient in shrimp diets, mainly due to the inclusion of fishmeal (Oujifard et al, 2012). The quality of fishmeal is determined by several factors, including the type of fish used, processing method and storage conditions. Good quality meals are usually obtained from fish with a high nutritional value, such as sardines, anchovies, and tuna.
In addition to quality factors, the availability of fishmeal is extremely limiting, as it is a finite ingredient that depends on the renewal capacity of the schools, that is, it is limited and unsustainable, obtained from predatory fishing that in most cases occurs, does not respect the reproduction cycle of fish (Sookying et al, 2013). In this sense, sustainable resources become essential, with possibilities for growth and supply of the shrimp market in the long term.
Availability
It is possible to reduce the use of fishmeal by combining other less expensive proteins and with a combination of amino acids that meets the requirements of shrimp. Some alternatives are animal meal from the slaughterhouse of poultry, pigs and cattle, blood meal dried by spraying, yeast, proteins of bacterial origin, among others (Hu et al, 2013).
Meals of animal origin, such as meat and bone meal, contain high levels of methionine, which theoretically becomes a good alternative to replace fishmeal, but it is a product whose use is prohibited in animal diets in some countries of the European Union, in addition to presenting some problems of palatability and contaminants (Sweetman, 2008).
Plant proteins, such as soybean meal, are used in fish and shrimp feed as an alternative source of protein. However, attention should be paid to anti-nutritional factors, which can adversely affect animal performance, such as: protease and tannin inhibitors, anti-vitamins, and other substances, such as phytoestrogens (Francis, Makkar & Klaus 2001).
Currently, yeasts represent an important source of protein capable of replacing (totally or partially) fishmeal (Alloul et al, 2021). It is an ingredient with wide availability and competitive prices. In Brazil, only 10% of the total produced is currently used for animal feed, of an amount of 1 million tons, only 100 thousand tons are used (UNICA, 2023).
Nutritional characteristics
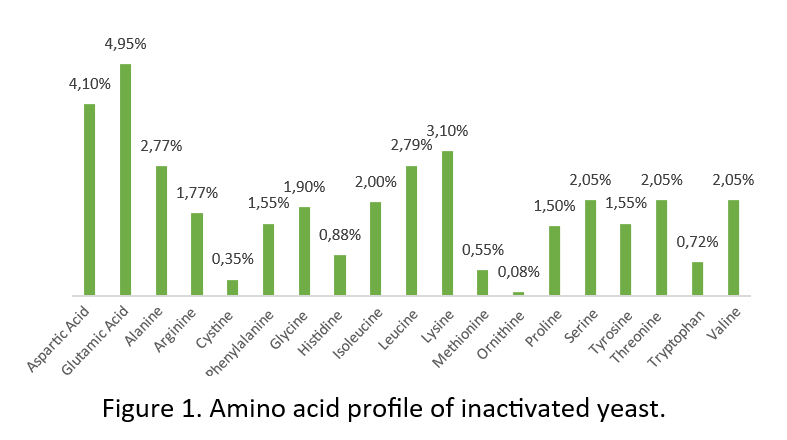
Yeasts are a well-known product in aquaculture (whole or fractionated) as a source of amino acids (Figure 1), proteins, minerals and vitamins, with a positive effect on shrimp growth and immunity (Gamboa-Delgado et al. 2016; Álvarez-Sánchez et al. 2018). Another point is the presence of glutamic acid and aspartic acid, which are related to the process of attractiveness and palatability in diets (Heu et al, 2003).
The use of yeast varies according to its processing. When only inactivated and dried, yeasts are used as a protein source and can provide up to 45% protein with a digestibility close to 70%. If processed, in the case of autolysis it can reach 80% and if hydrolyzed it exceeds 80% (Rostagno et al, 2017). There are also cases where you only want to use it as a functional food, as they are sources of β-glucans and mannanoligosaccharides, which in turn help strengthen the immune system and bind to pathogens, respectively (Chi et al, 2006).
Sustainability and Environment
Yeasts play an important role in the control and resistance to various diseases. According to Ceseña et al. (2021) they can be used, in addition to their nutritional value, as antioxidants and immunostimulants, which allows reducing the use of antibiotics in commercial livestock.
Another point is the sustainability of the product in its production process and with the environment. The yeasts from the manufacture of ethanol are inserted in an activity with negative carbon production and that has managed to reduce by more than 630 million tons of CO2eq since March 2022 in Brazil, that is, to achieve this CO2 economy it would be necessary to plant more than 4,500 million native trees in the next 20 years (UNICA, 2023).
It is also a product that allows to reduce the use of antibiotics, medicines, and other chemicals, since it does not affect the aquatic environment. Water quality is a very important point to understand and control diseases and pathogens, in addition to avoiding environmental pollution, either with molecules that degrade the environment or microorganisms, or affecting biodiversity. Yeasts are already part of the environment or microbiota of aquatic organisms and can improve animal welfare and health, influencing the microecological environment (Deng et al, 2013). The same authors observed that with the use of Saccharomyces cerevisiae directly in the water reduced the amount of NH3, chemical oxygen demand and sulfites in the water and sediments of the lagoon.
Immune system and disease prevention
The use of yeasts and their products represents a possible and viable disease prevention and control strategy to improve the quality and sustainability of aquaculture production. Several studies (Burgents et al. 2004; Zhenming et al. 2006: Sajeevan et al. 2009a,b; Bai et al. 2010, 2014; Babu et al. 2013; Deng et al. 2013; Sang et al. 2014; Wilson et al. 2015; Neto and Nunes 2015; Jin et al. 2018), demonstrated beneficial effects on components of the shrimp immune system, such as: effects on anticoagulant proteins, agglutination of pathogens, antioxidant effect, improvement in humoral components, lysozyme activity, phagocytosis processes, among others.
Some studies (Felix et al. 2008; Guo et al. 2016) have demonstrated the efficacy of the use of yeast under vibriosis conditions on shrimp performance and against myonecrosis virus (IMNV) and white spot virus (WSSV) in L. vannamei (Sukumaran et al. 2010; Zhu et al, 2010; Neto & Nunes, 2015; Wilson et al. 2015). These results demonstrate the immunostimulatory activity of β-glucans 1,3 and 1,6 supplemented with yeast. Immunostimulants may increase phagocytosis of pathogens by activating phagocytic cells in hemolymph, increasing the antibacterial and antiseptic properties of hemolymph, activating the PROPO system, and mediating recognition and phagocytosis signals (Bondad-Reantaso et al. 2005; Castex et al. 2010).
Alongside the β-glucans in the yeast cell wall are also mannanoligosaccharides (MOS). They are a complex of indigestible carbohydrates that prevent the adhesion of pathogenic bacteria because it acts by blocking the adhesion of microbial lectins to carbohydrates present on the surface of intestinal cells. Non-adherence to the intestinal membrane inhibits colonization of the infectious pathogen, which is eliminated in feces (Genc et al. 2007; Zhang et al. 2012; Genc and Ebeoglu 2013; Sang et al. 2014).
All these mechanisms, in addition to providing productive improvements (Chi et al, 2016), also allow a reduction in animal mortality (Nimrat et al, 2019) resulting from the observed benefits and the responsiveness of animals to challenges.
Final considerations
The use of yeasts in shrimp feed is very solid in the nutritional and functional sense, that is, they are an alternative source to fishmeal, with long-term supply and supply potential, as well as being sustainable. They allow to reduce the use of antibiotics and other medicines since they have a direct effect modulating the immune system and agglutinating pathogens, in addition to not damaging the aquatic environment.
References
Alloul, A.; Wille, M.; Lucenti, P.; Bossier, P.; Van Stappen, G.; Vlaeminck, S.E. Purple Bacteria as Added-Value Protein Ingredient in Shrimp Feed: Penaeus vannamei Growth Performance, and Tolerance against Vibrio and Ammonia Stress. Aquaculture 2021, 530, 735788.
Alvarez-Sanchez, A.R., Nolasco, H., Pena, A., Mejia, H. In vitro digestibility of Yarrowia lipolytica yeast and growth performance in whiteleg shrimp Litopenaeus vannamei. J Fish Aquat Sci, 18 (2018).
Babu, D. T., Antony, S. P., Joseph, S. P., Bright, A. R., & Philip, R. (2013). Marine yeast Candida aquaetextoris S527 as a potential immunostimulant in black tiger shrimp Penaeus monodon. Journal of invertebrate pathology, 112(3), 243-252.
Bai, N., Zhang, W., Mai, K., Wang, X., Xu, W., & Ma, H. (2010). Effects of discontinuous administration of β-glucan and glycyrrhizin on the growth and immunity of white shrimp Litopenaeus vannamei. Aquaculture, 306(1-4), 218-224.
Bondad-Reantaso, M. G., Subasinghe, R. P., Arthur, J. R., Ogawa, K., Chinabut, S., Adlard, R., … & Shariff, M. (2005). Disease and health management in Asian aquaculture. Veterinary parasitology, 132(3-4), 249-272.
Burgents, J. E., K. G. Burnett, and L. E. Burnett. 2004. Disease resistance of Pacific white shrimp, Litopenaeus vannamei, following the dietary administration of a yeast culture food supplement. Aquaculture 231:1–8. doi:10.1016/j.aquaculture.2003.09.003.
Castex, M., Lemaire, P., Wabete, N., & Chim, L. (2010). Effect of probiotic Pediococcus acidilactici on antioxidant defences and oxidative stress of Litopenaeus stylirostris under Vibrio nigripulchritudo challenge. Fish & shellfish immunology, 28(4), 622-631.
Ceseña, C.E.; Vega-Villasante, F.; Aguirre-Guzmán, G.; Luna-Gonzalez, A.; Campa-Cordova, A.I. Update on the use of yeast in shrimp aquaculture: A mini review. Int. J. Aquat. Res. 2021, 13, 1–16.
Chi, Z., Z. Liu, L. Gao, F. Gong, C. Ma, X. Wang, and H. Li. 2006. Mine yeasts and their applications in mariculture. Journal of Ocean University of China 5:251–56. doi:10.1007/s11802-006-0010-5.
Deng D, Mei C, Mai K, Tan BP, Ai Q, Ma H (2013) Effects of a yeast-based additive on growth and immune responses of white shrimp, Litopenaeus vannamei (Boone, 1931), and aquaculture environment. Aquaculture Research 44: 1348–1357.
Felix, W., Oliveira, C., Moreira, R. A., Teixeira, J. A., & Domingues, L. (2008). Expression of frutalin, an α-D-galactose-binding jacalin-related lectin, in the yeast Pichia pastoris. Protein Expression and Purification, 60(2), 188-193.
Francis G., Makkar H.P.S. & Klaus B. (2001) Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture, 199, 197–227.
Gamboa-Delgado, J., Navarro, Y.I.M., Nieto-Lopez, M.G., Villarreal-Cavazos, D.A., Cruz-Suarez, L.E. Assimilation of dietary nitrogen supplied by fish meal and microalgal biomass from Spirulina (Arthrospira platensis) and Nannochloropsis oculata in shrimp Litopenaeus vannamei fed compound diets. J Appl Phycol, 31 (2019), pp. 2379-2389.
Genc, M. A., Yilmaz, E., Genc, E., & Aktas, M. (2007). Effects of dietary mannan oligosaccharides (MOS) on growth, body composition, and intestine and liver histology of the hybrid tilapia (Oreochromis niloticus x 0. aureus). Israeli Journal of Aquaculture-Bamidgeh, 59.
Genc, M. A., & Ebeoglu, B. (2013). The effects of different salinity and supplemented mannan oligosaccharides (MOS) on growth of Litopenaeus varmamei (Penaeus: Decapoda). J. Anim. Veterin. Adv, 12(9), 942-947.
Guo, Y., Zhang, B., Wang, W., Li, Z., Han, Q., D’inca, R. (2016). Dietary live yeast and mannan-oligosaccharide supplementation attenuate intestinal inflammation and barrier dysfunction induced by Escherichia coli in broilers. British Journal of Nutrition, 116(11), 1878-1888.
Heu, M.-S.; Kim, J.-S.; Shahidi, F. Components and nutritional quality of shrimp processing by-products. Food Chem. 2003, 82, 235–242
Hu, L., Yun, B., Xue, M., Wang, J., Wu, X., Zheng, Y., Han, F., 2013. Effects of fish meal quality and fish meal substitution by animal protein blend on growth performance, flesh quality and liver histology of Japanese seabass (Lateolabrax japonicus). Aquaculture 372-375, 52–61.
Jin, W., Yang, K., Barzilay, R., & Jaakkola, T. (2018). Learning multimodal graph-to-graph translation for molecular optimization. arXiv preprint arXiv:1812.01070.
Li, W., Lu, H., He, Z., Sang, Y., & Sun, J. (2021). Quality characteristics and bacterial community of a Chinese salt-fermented shrimp paste. LWT, 136, 110358.
Nimrat, S., Khaopong, W., Sangsong, J., Boonthai, T., & Vuthiphandchai, V. (2021). Dietary administration of Bacillus and yeast probiotics improves the growth, survival, and microbial community of juvenile whiteleg shrimp, Litopenaeus vannamei. Journal of Applied Aquaculture, 33(1), 15-31.
Oujifard A, Seyfabadi J, Kenari AA, Rezaei M. 2012. Growth and apparent digestibility of nutrients, fatty acids and amino acids in Pacific white shrimp, Litopenaeus vannamei, fed diets with rice protein concentrate as total and partial replacement of fish meal. Aquaculture 342-343:56e61.
Rostagno, H.S., Albino, L.F.T., Hannas, M.I., Donzele, J.L., Sakomura, N.K., Perazzo, F.G., Saraiva, A., Teixeira, M.L., Rodrigues, P.B., Oliveira, R.F., Barreto, S.L. Oliveira Brito, C. Tabelas Brasileiras para Aves e Suínos. H.S. Rostagno (Ed.), Composiçao de Alimentos e Exigencias Nutricionais (4th ed.), Universidade Federal de Viçosa, Viçosa, Brazil (2017).
Sabry Neto, H., Santaella, S. T., & Nunes, A. J. P. (2015). Bioavailability of crude protein and lipid from biofloc meals produced in an activated sludge system for white shrimp, Litopenaeus vannamei. Revista Brasileira de Zootecnia, 44, 269-275.
Sajeevan, T.P., Rosamma Philip, I.S. Bright Singh, Dose/frequency: A critical factor in the administration of glucan as immunostimulant to Indian white shrimp Fenneropenaeus indicus, Aquaculture, Volume 287, Issues 3–4, 2009, Pages 248-252, ISSN 0044-8486, https://doi.org/10.1016/j.aquaculture.2008.10.045.
Sajeevan, T.P., Lowman, D.W., Williams, D.L., Selven, S., Anas, A. and Rosamma, P. (2009), Marine yeast diet confers better protection than its cell wall component (1-3)-β-d-glucan as an immunostimulant in Fenneropenaeus indicus. Aquaculture Research, 40: 1723-1730. https://doi.org/10.1111/j.1365-2109.2009.02275.x
Sang, X., Oni, A. A., & LeBeau, J. M. (2014). Atom column indexing: atomic resolution image analysis through a matrix representation. Microscopy and Microanalysis, 20(6), 1764-1771.
Sookying, D.; Davis, D. A. and Silva, F.S.D. 2013. A review of the development and application of soybean-based diets for Pacif white shrimp Litopenaeus vannamei. Aquaculture Nutrition 19:441-448. doi: 10.1111/anu.12050.
Sukumaran, V., Lowman, D. W., Sajeevan, T. P., & Philip, R. (2010). Marine yeast glucans confer better protection than that of baker’s yeast in Penaeus monodon against white spot syndrome virus infection. Aquaculture Research, 41(12), 1799-1805.
Sweetman J. (2008) The role of yeast derived proteins in modern aqua feed formulations. International AquaFeed, 8, 19–23.
UNICA – União da Indústria de Cana de Açúcar. https://unicadata.com.br/. 01/09/2023.
Zhang, G. L., Ma, X., X. Fan, P., Li, L. S., Qiao, S. Y. & Li, D. F. (2012). Butyrate promotes the recovering of intestinal wound healing through its positive effect on the tight junctions. Journal of animal science, 90(suppl_4), 266-268.
Zhenming, C., Zhiqiang, L., Lingmei, G. et al. Marine yeasts and their applications in mariculture. J Ocean Univ. China 5, 251–256 (2006). https://doi.org/10.1007/s11802-006-0010-5.
Zhu XZ, Liu YJ, Tian LX, Mai KS, Zheng SX, Pan QJ et al. (2010) Effects of dietary protein and lipid levels on growth and energy productive value of pacific white shrimp, Litopenaeus vannamei, at different salinities. Aquaculture Nutrition 16: 392–399.
Wilson, M. J., Frickel, S., Nguyen, D., Bui, T., Echsner, S., Simon, B. R., … & Wickliffe, J. K. (2015). A targeted health risk assessment following the Deepwater Horizon oil spill: polycyclic aromatic hydrocarbon exposure in Vietnamese-American shrimp consumers. Environmental health perspectives, 123(2), 152-159.





















